Resources




Innovamol uses cutting-edge tools to provide top-notch services, including:


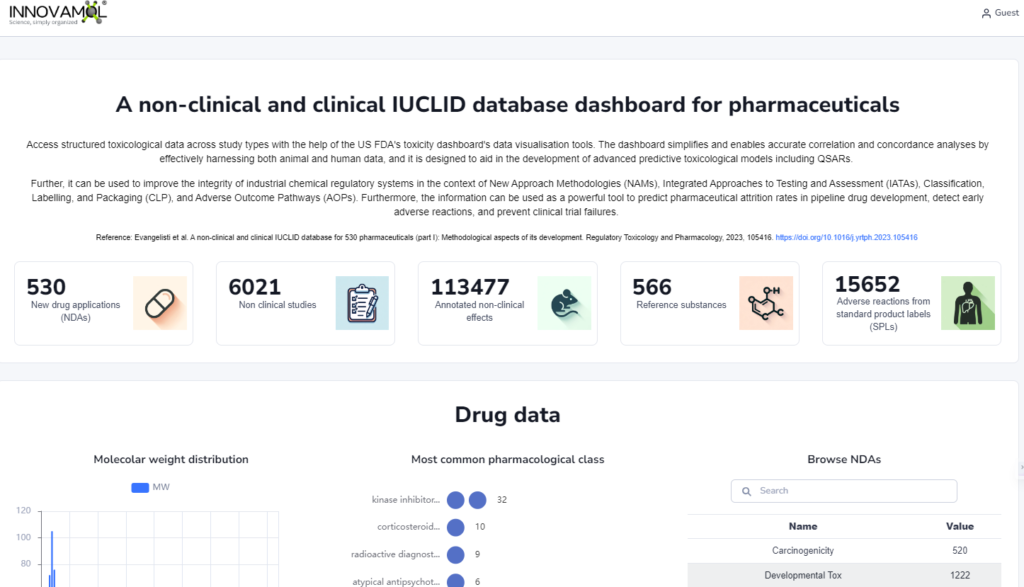
Access structured toxicological data across study types.
The dashboard simplifies and enables accurate correlation and concordance analyses by effectively harnessing both animal and human data, and it is designed to aid in the development of advanced predictive toxicological models including QSARs. Further, it can be used to improve the integrity of industrial chemical regulatory systems in the context of New Approach Methodologies (NAMs), Integrated Approaches to Testing and Assessment (IATAs), Classification, Labelling, and Packaging (CLP), and Adverse Outcome Pathways (AOPs). Furthermore, the information can be used as a powerful tool to predict pharmaceutical attrition rates in pipeline drug development, detect early adverse reactions, and prevent clinical trial failures.

Streamline Literature Reviews and Rapid Reviews
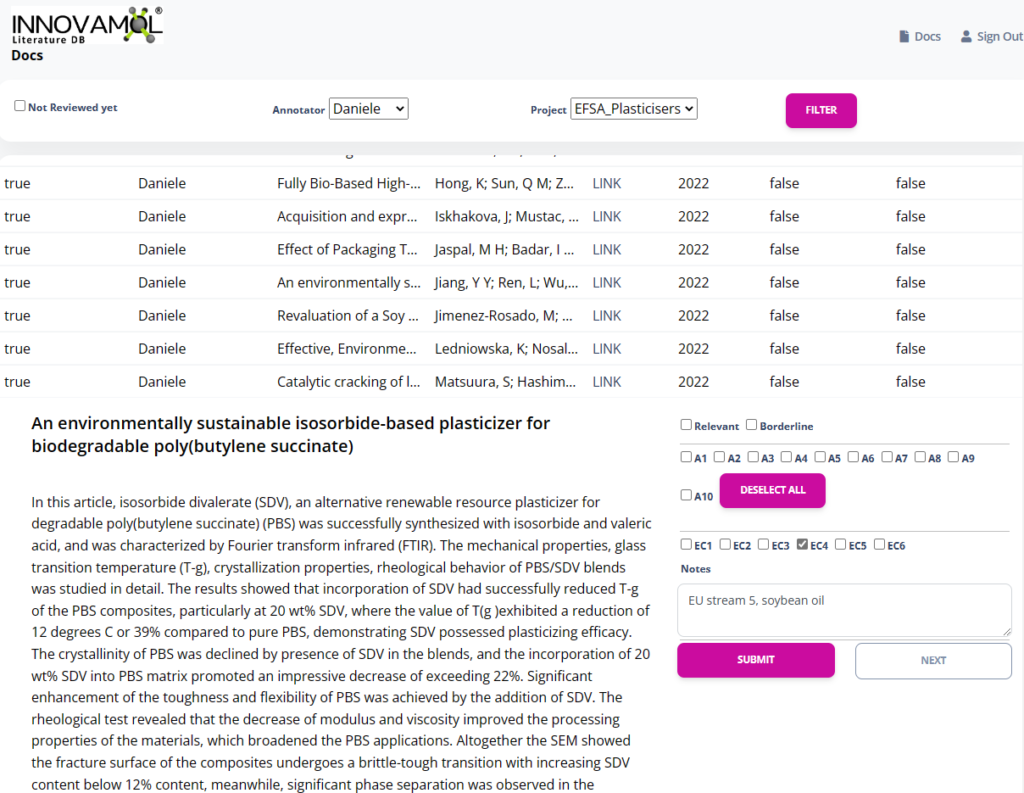
InnoLiterature® database is a powerful, web-based tool designed to simplify and streamline extensive literature review and rapid review projects conducted within our organization. Developed in-house, this user-friendly platform enables our team members to work simultaneously and seamlessly, ensuring transparency, reproducibility, and efficiency. Utilizing the framework of PROSPERO, the database facilitates organizing, annotating, and extracting data with ease, while also supporting compatibility with RIS files and other protocols (e.g., Cochrane). Experience the benefits of a clear and consistent approach to managing your scientific review projects as we harness the power of InnoLiterature® database to deliver exceptionally accurate and rapid results for your organization.

Optimize Chemical Information management
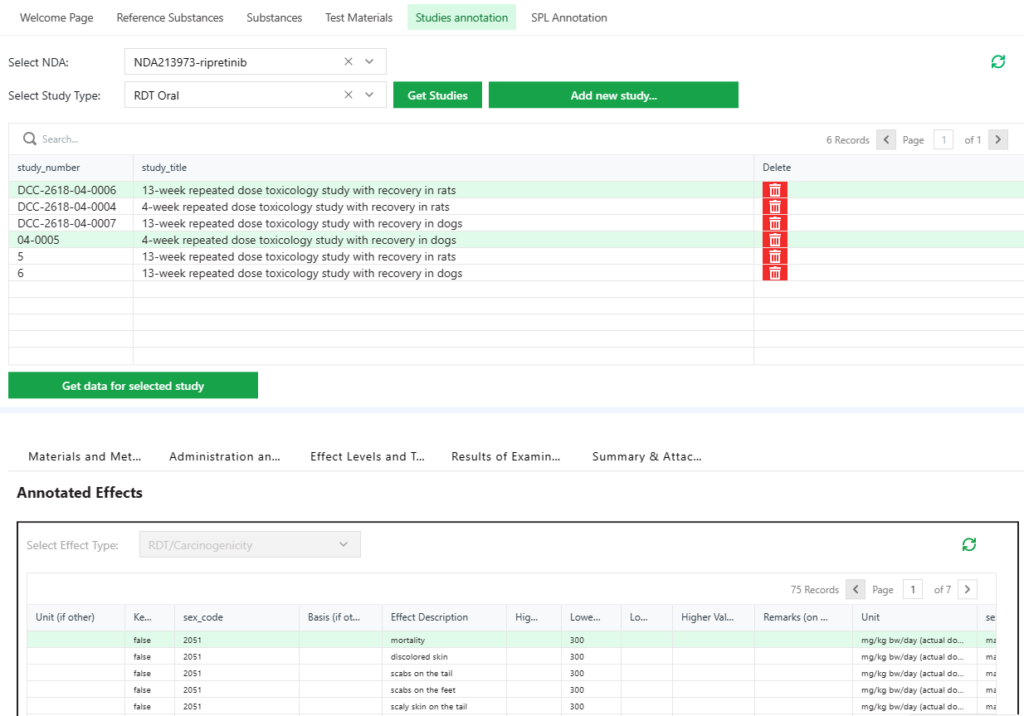
The InnoCLID® database is our in-house developed, web-based tool designed to optimize the process of capturing, organizing, and managing chemical information in IUCLID format. This innovative platform enables efficient compilation of chemical data from various sources, including toxicity data, non-clinical animal studies, and human information, streamlining data analysis. InnoCLID® database not only enhances scientific and regulatory knowledge but also supports the development of safe-by-design approaches and quantitative structure-activity relationship (QSAR) or mechanistic studies. Experience a more efficient approach to managing and organizing your chemical data with the capabilities of the InnoCLID ® Database.
A part of the data contained in the database can be easily visualized throught the FDA IUCLID DataViz: https://toxicity-dataviz.innovamol.com

Enhanced Data Organization in interdisciplinary science
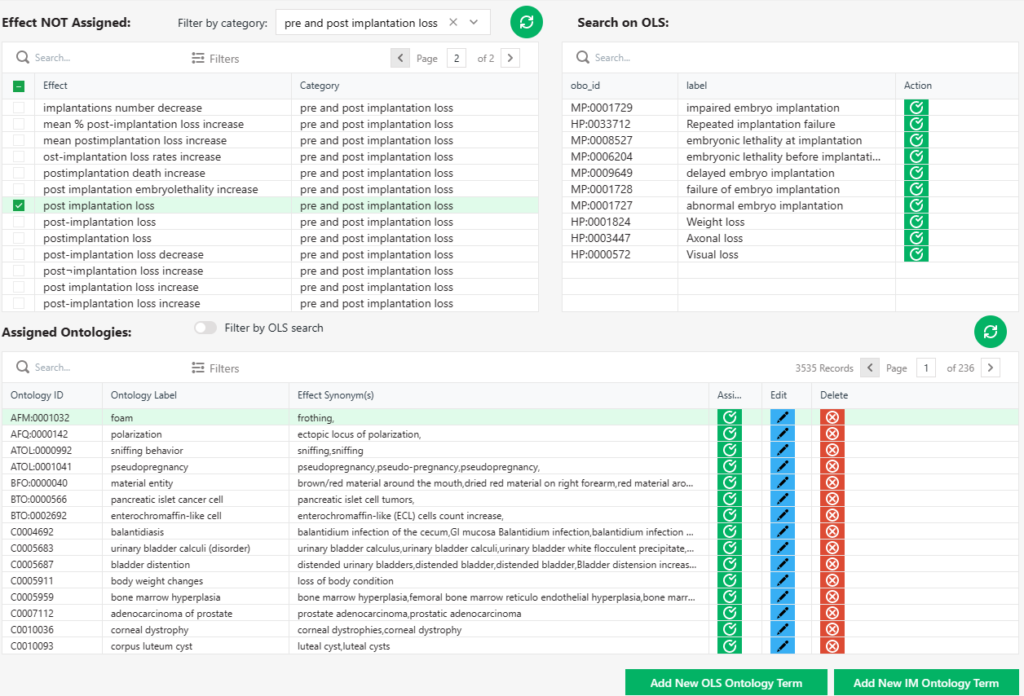
The InnoOntology® database is an innovative, web-based tool that significantly improves the way scientific data is organized and managed. Developed in-house, this advanced platform effectively captures and stores ontology information, ensuring enhanced data organization, standardized terminology, and cross-referencing capabilities. By facilitating interdisciplinary research, InnoOntology® database bridges the gap between various scientific disciplines and provides a common language and framework for seamless collaboration. Experience the benefits of accurate, consistent, and easily integrated data as InnoOntology® database supports machine learning, AI-driven insights, and new discoveries in the realm of scientific data analysis.
One ontology that we recently developed thanks to this tool is the Preclinical and Clinical Concept Ontology (PaCCO) https://github.com/innovamol/PaCCO